Diy soxhlet extractor1/1/2024  
You are supposed to dry it better, but I'm only playing and I didn't want to wait any longer or degrade it with heat. I let the material air dry for a few days. Again if you go with the PYREX® version you can pay $90 for the size I have, crazy! Probably worth it if is a profession and you are dealing with rare compounds, but for a hobby. My setup has a fritted disc extraction thimble. The consistent and known particle retention is useful for repeatability. Cellulose thimbles are made specifically for this purpose. The material can be packaged in a coffee filter. I've seen people with small extractors use a cotton ball packed into the bottom. The main purpose of the thimble is to prevent the solid from getting drawn out of the Soxhlet extractor during siphon. The process starts by packing the source material into the thimble. Sohlext extraction is a reflux operation where the volatiles are cooled and not allowed to escape. They consist of 4 main parts, an Allihn condenser, the actual Soxhlet extractor, a thimble of some sort, and a boiling flask.Ī Sohxlet extractor is effectively a closed system which allows extractions to be run for hours or days unattended. If you don't mind slow boating one form China they are an order of magnitude cheaper. They can cost you an easy grand if you buy a nice PYREX® setup from Corning. A guy like me can only read so many papers talking about them before the urge to get one grows too high to resist. I went for a solvent extraction system more specifically, a Soxhlet extractor. A downside is that it's easy to spend thousands of dollars on the equipment needed. Since the boiling point of CO 2 is -78.5☌, it's trivial to boil it all off after extraction. One paper I've read found that CO 2 the region right about 30˚C and a pressure right below 300 bar gave some of the highest yields when extracting basil. Varying the exact temperature and pressure will change the solubility of various compounds. 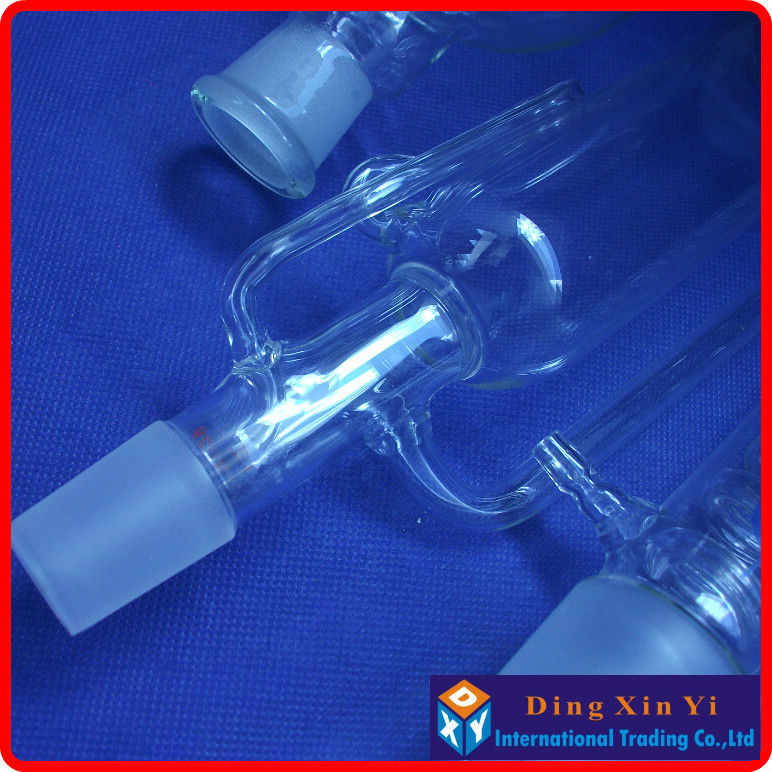
Yeah thermodynamics! In this region there is no distinct liquid and gas phase. A supercritical fluid is a substance at a temperature and pressure above the critical point on its phase diagram. Possibly the coolest extraction method is supercritical fluid extraction. Plus it feels like real chemistry and you can get hurt! Supercritical fluid extraction (SFE!) The advantage is that you will generally get the the highest yields of the desired compound. Depending on your goal, many of these compounds are undesired. You will get essential oils, esters and heavy terpenes, fatty acids, triglycerides, waxes, resins, and some pigments. More components of plant matter are soluble in these solvents than in water. Solvents like chloromethane, dichloromethane, trichloromethane, ethoxyethane, methanol, ethanol, petroleum ether, butane, and hexane are common. After all, water is the universal solvent! In this case I'm referring more to non-polar organic solvents (the ones with a low dielectric constant for an electronics reference). Technically hydrodistillation is a solvent extraction. 
Many compounds degrade at temperatures this high. One downside of hydrodistillation is that water doesn't boil until 100˚C. One advantage of using water is that your product will contain a lot less impurities due to their lower solubility. This is a mixture of the components that are water soluble. A third layer of hydrosols will often form. The vapor is condensed two layers form and can be separated by decantation. 
When heated, the two liquids form an azeotrope and boil off together. Hydrodistillation takes two main forms, one where the material to be extracted is mixed into the water, and one where steam is passed through a bed of the material. There are a handful of ways that I'm familiar with for extracting oils, volatiles, and other compounds of interest from solid materials. Rather than take 5 minutes to replant I decided it was time to blow all of my disposable income on some glass labware I'd wanted for years and extract the good stuff! It had started to yellow & was in serious need of a larger pot. Recently I had some basil growing in the kitchen that I wasn't using fast enough. At times I've had tomato plants going so well in one of my hydroponic setups that I couldn't reach them all! That became a mess. They are fun to take care of and a cheap source of food and ornamentals. For years, depending on my living situation, I've kept an indoor garden. There is something old–timey and satisfying about being the source of your own stuff. I've always had a bit of a maker or DIY lean in my hobbies. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
